Compliance Without Compromise: Accelerating Innovation Safely
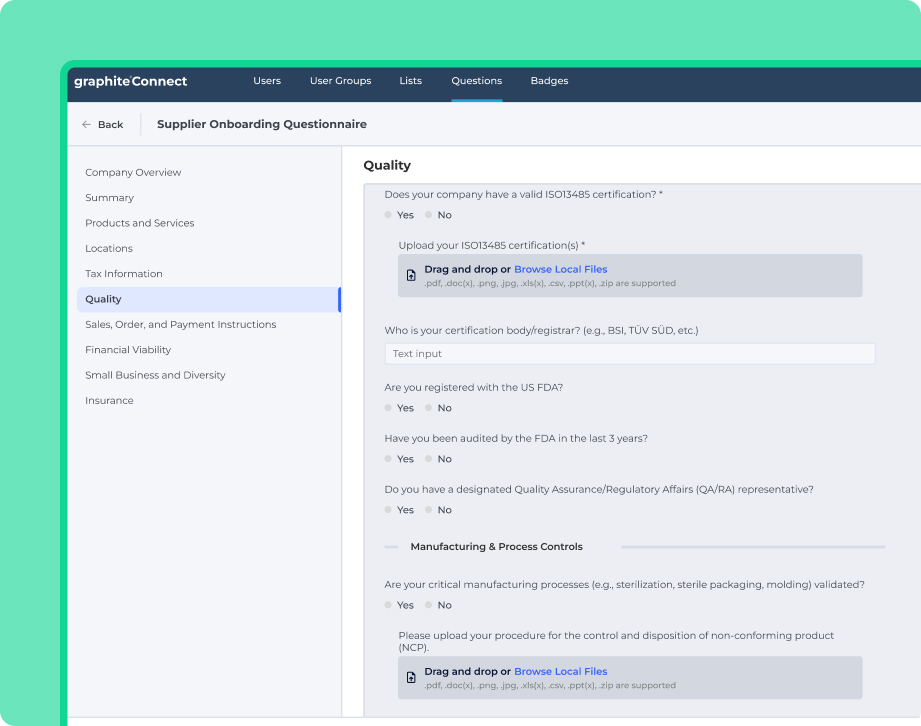
In the highly regulated Pharma and Med Device industries, every minute spent on manual supplier vetting, document chasing, or risk assessment delays life-saving products.
Graphite provides the single, validated source of truth your procurement, quality, and compliance teams need to move with unprecedented speed and confidence.
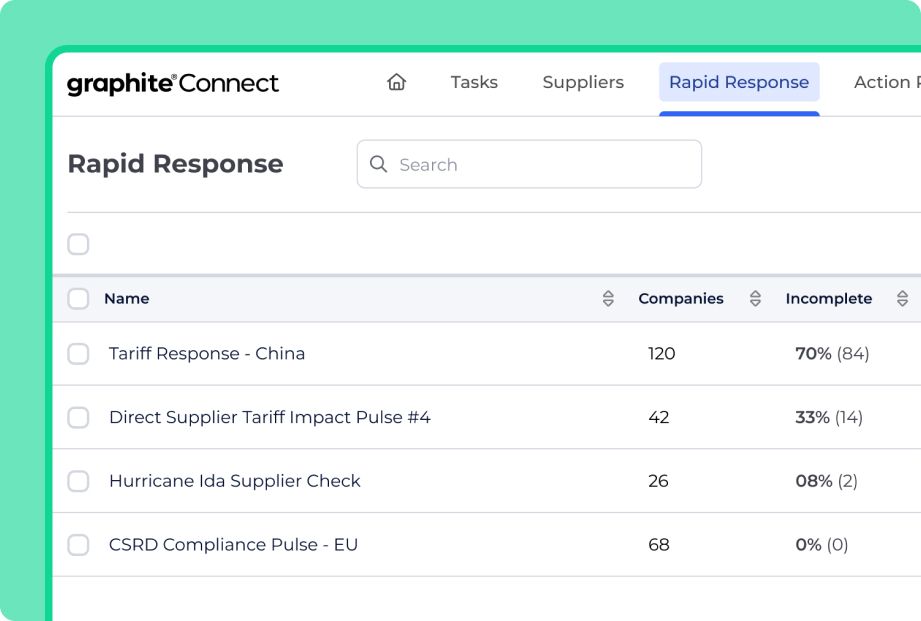
Fast Supplier Collaboration to Deliver GxP Excellence

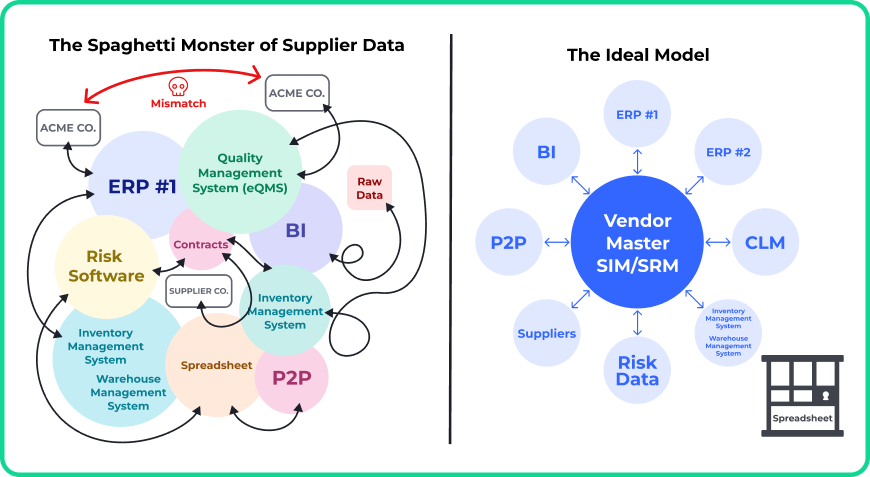
Bad supplier data is holding you back.
In our 2026 State of Supplier Data white paper, learn the key challenges in building a supplier master data management strategy and how it’s killing the ROI on your AI investments.
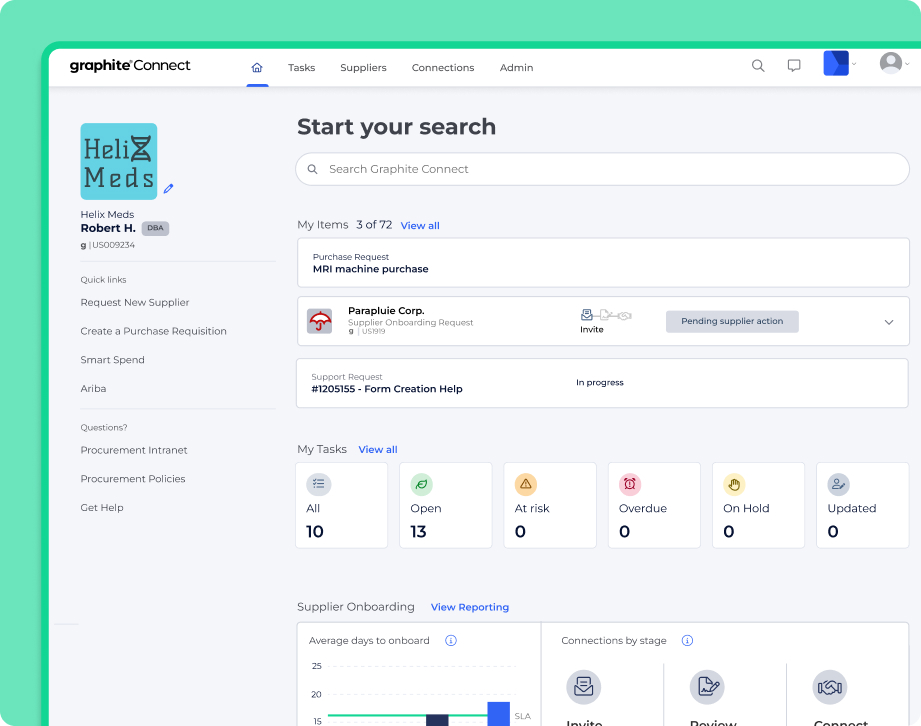
Unlock total transparency for stakeholders
Every step of your process is clear to the teams following the project’s success and delivery timelines. Quality, Compliance, Legal, Supply Chain teams know who is next up to bat in reviewing and approving suppliers, keeping you on track and preventing project delays.